
Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses theĬreative Commons Attribution-NonCommercial-ShareAlike License Although the equilibration is slow at neutral pH, it is catalyzed by both acid and base. Mutarotation occurs by a reversible ring-opening of each anomer to the open-chain aldehyde, followed by reclosure. Called mutarotation, this change in optical rotation is due to the slow interconversion of the pure anomers to give the same 37 : 63 equilibrium mixture. That is, the specific rotation of the α-anomer solution decreases from +112.2 to +52.6, and the specific rotation of the β-anomer solution increases from +18.7 to +52.6. When a sample of either pure anomer is dissolved in water, however, its optical rotation slowly changes until it reaches a constant value of +52.6. Pure α- D-glucopyranose has a melting point of 146 ☌ and a specific rotation D = +112.2 pure β- D-glucopyranose has a melting point of 148 to 155 ☌ and a specific rotation D = +18.7. 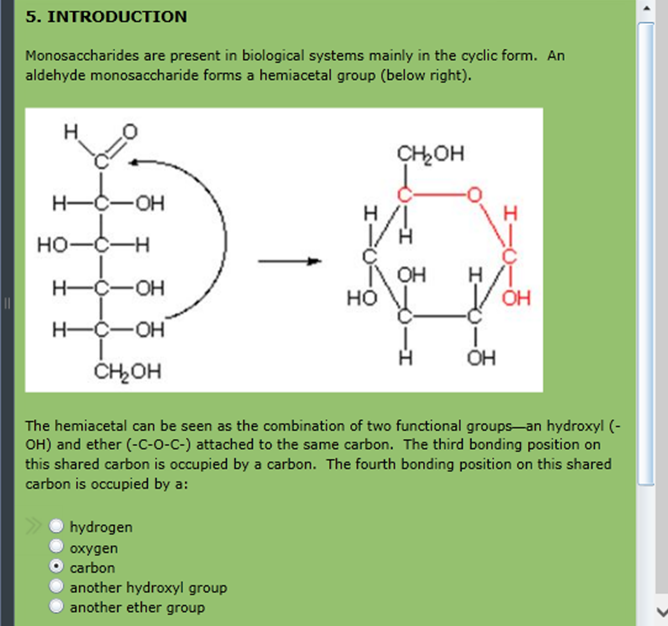
The two pyranose anomers result from addition of the C6 –OH group to the C2 carbonyl the two furanose anomers result from addition of the C5 –OH group to the C2 carbonyl.īoth anomers of D-glucopyranose can be crystallized and purified. Thus, β- D-glucopyranose is the least sterically crowded and most stable of the eight D aldohexoses.įigure 25.6 Pyranose and furanose forms of fructose in aqueous solution. Note that in β- D-glucopyranose, all the substituents on the ring are equatorial. The compound with its newly generated –OH group trans to the –OH at the lowest chirality center is called the β anomer its full name is β- D-glucopyranose. The compound with its newly generated –OH group at C1 cis to the –OH at the lowest chirality center in a Fischer projection is called the α anomer its full name is α- D-glucopyranose. For example, glucose cyclizes reversibly in aqueous solution to a 37 : 63 mixture of two anomers ( Figure 25.5). The hemiacetal carbon atom is referred to as the anomeric center. When an open-chain monosaccharide cyclizes to a pyranose form, a new chirality center is generated at the former carbonyl carbon and two diastereomers, called anomers, are produced. For D sugars, the terminal –CH 2OH group is on the top of the ring, whereas for L sugars, the –CH 2OH group is on the bottom. Note that an –OH group on the right in a Fischer projection is on the bottom face of the pyranose ring, and an –OH group on the left in a Fischer projection is on the top face of the ring. 
By convention, the rings are usually drawn by placing the hemiacetal oxygen atom at the right rear, as shown in Figure 25.5. Like cyclohexane rings ( Section 4.6), pyranose rings have a chairlike geometry with axial and equatorial substituents. The name pyranose is derived from pyran, the name of the unsaturated six-membered cyclic ether. Glucose, for instance, exists in aqueous solution primarily in the six-membered pyranose form resulting from intramolecular nucleophilic addition of the –OH group at C5 to the C1 carbonyl group ( Figure 25.5). Five- and six-membered cyclic hemiacetals are relatively strain-free and particularly stable, and many carbohydrates therefore exist in an equilibrium between open-chain and cyclic forms. If the carbonyl and the hydroxyl group are in the same molecule, an intramolecular nucleophilic addition can take place, leading to the formation of a cyclic hemiacetal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
